|
elements having the same number of electrons). This theory fails to account for isoelectronic species (i.e.If the electron pairs are separated by a large distance, the repulsions between them are reduced, and the energy of the molecule is reduced.As a result, the energy of the molecules increases. If electron pairs around the core atom get too close, they will reject each other. 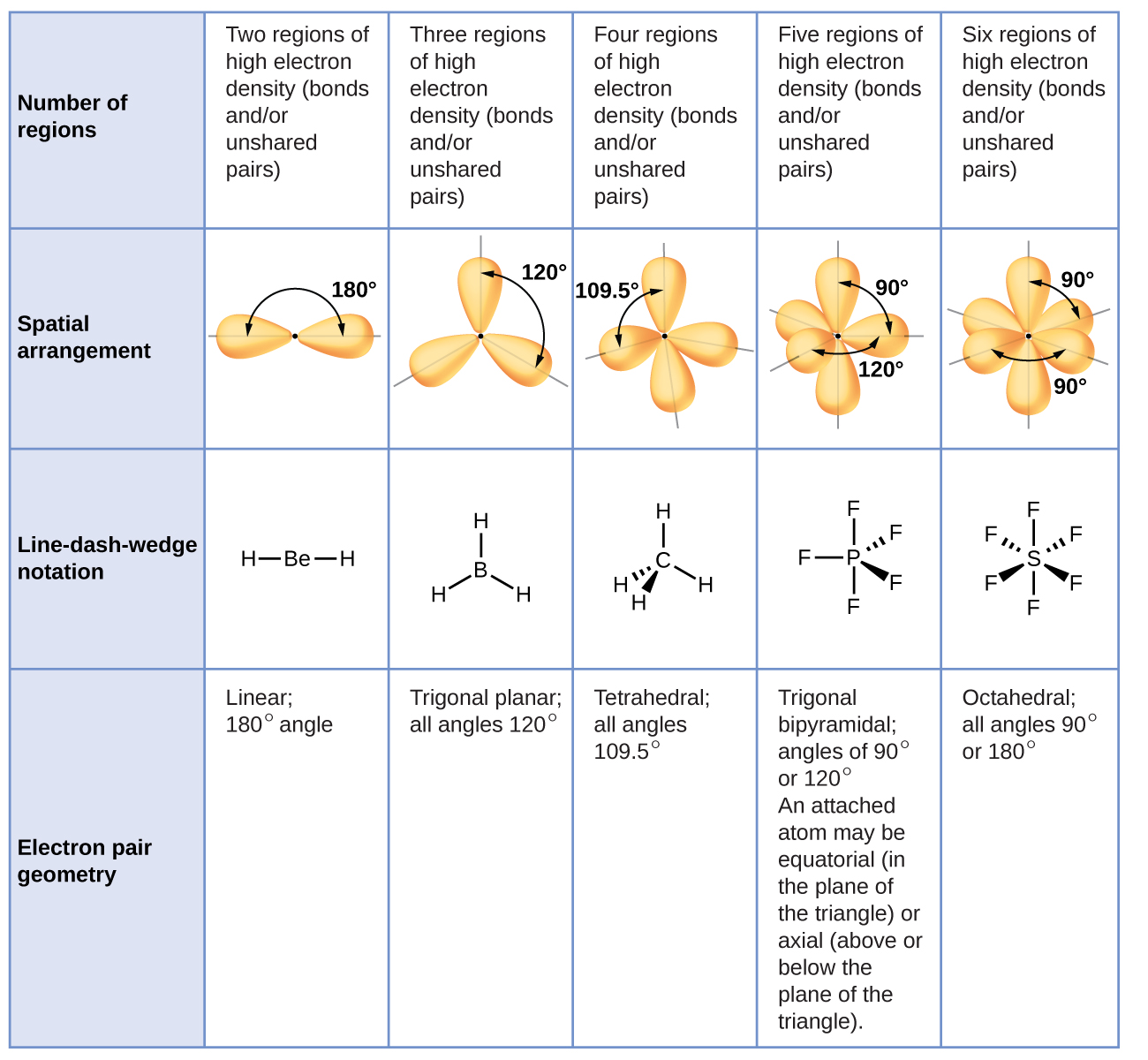
The repulsion is largest between two lone pairs and smallest between two bond pairs.The VSEPR theory can be applied to any molecule’s resonance structure.If the centre atom is surrounded by both lone pairs and bond pairs of electrons, the molecule will be deformed.If the core atom of the molecule is surrounded by electron bond pairs, an asymmetrically structured molecule can be expected.The valence shell can be visualised as a sphere with electron pairs clustered on its surface to reduce the distance between them.Electron pairs have a tendency to organise themselves in such a way that their electron-electron repulsion is minimised while their distance is increased.The total amount of valence shell electron pairs determines the molecule’s structure.In polyatomic molecules (molecules composed of three or more atoms), one of the constituent atoms is designated as the central atom, to which all other atoms in the molecule are attached.Augmented Assignment Operators in Python. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
